TITRATIONSA series of three videos that cover
Written by: Simon Boman
Created: 3 Sep 2013 Edited: 18 Oct 2018. Grammatical changes made, removed absolute & relative uncertainties calculations to simplify legibility of the analysis section. |
|
Determining the concentration of ammonia in cloudy ammonia
Aim
To determine the concentration of ammonia (as NH₃) in the product 'cloudy ammonia' to verify the claim of 20g/L (note: this value is different to the one used in the videos above).
Introduction
"Cloudy ammonia" is a household cleaning product that claims to have an ammonia (NH₃) content of 20g/L, which is to be verified in the laboratory. The method chosen to determine this quantity is an acid-base titration due to the nature of Ammonia, being a proton acceptor, is known as an weak alkali, and hence requires an acid for titration. The acid chosen for this task was hydrochloric acid (HCl), however due to its low molecular weight, highly corrosive and hygroscopic properties, it is not an ideal chemical to be made as a standard solution. For this task, the HCl was standardised (titrated against) with a Na₂CO₃ standard solution, whose molecular weight of 106g/mol is greater than that of HCl whose molecular weight is 36.5g/mol, reducing relative errors in measurement during the weighing process, as well as its reduced hygroscopic properties limiting the affects of moisture on the weight measured.
|
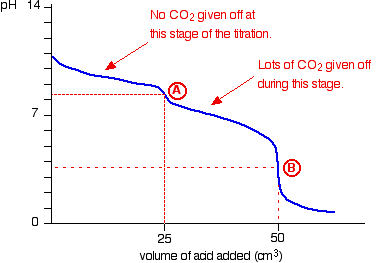
Figure 1: Hydrochloric Acid vs Sodium Carbonate pH Curve (Clark, 2002)
|
The indicator chosen was Methyl Orange, whose end point occurs between pH 3.1 - 4.4 (Clark, 2002), which closely matches the second equivalence point of the titration (pH) curve shown in figure 1 between a strong acid and a weak base. This equivalence point occurs at approximately pH 3-4, of the second stage of the chemical reaction between Hydrochloric Acid and Sodium Carbonate.
Stage 1: First equivalence point
Na₂CO₃ (aq) + HCl (aq) → NaHCO₃ (aq) + NaCl (aq) Stage 2: Second equivalence point NaHCO₃ (aq) + HCl (aq) →H₂O (l) + NaCl (aq) + CO₂ (g) |
Using the titration process, the concentration of the HCl solution can then be determined, and hence it can then be used as a standard against the cloudy ammonia. Due to the concentrated nature of the bottle of cloudy ammonia, a significant volume of HCl could be required (depending on the concentration of the HCl solution) which may exceed the volume capacity of the burette used in the experiment and hence will require dilution to attain an acceptable titre volume of approximately 20-30 mL of a 50mL burette.
|
Once the HCl solution has been standardised, this will then be used to titrate against the diluted cloudy ammonia solution to verify the concentration, and then convert this concentration as a mass per volume unit to compare with the labelling of the bottle of cloudy ammonia.
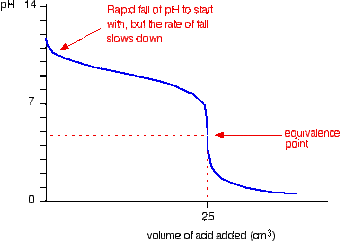
Methyl Orange indicator was used for this titration process as well since the equivalence point occurs at approximately the same pH range as Methyl Orange's end point of pH 3.1 - 4.4 (Clark, 2002) (see figure 2). |
Figure 2: Hydrochloric Acid vs Ammonia pH Curve (Clark, 2002)
|
Materials
|
|
Procedure
Preparation of Na₂CO₃ standard solution (stock)
A mass of approximately 1.06g was weighed and measured of anhydrous Na₂CO₃ and diluted to a volume of 250mL in a volumetric flask which was then inverted a number of times to mix. The concentration of this standard solution was then calculated to be used in the titration against HCl.
Standardisation of HCl solution
Before titration, the burette was rinsed with distilled water followed by another rinse with the HCl solution to ensure that the burette was clean and free of impurities as much as possible. This was then filled with the HCl solution.
Similarly, the pipette to be used to transfer aliquots of Na₂CO₃ was rinsed once with distilled water from a 50mL beaker and deposited down the sink before rinsing again with a small sample of Na₂CO₃ solution from another 50mL beaker. Once the pipette had been rinsed, a 20.0mL aliquot of Na₂CO₃ was transferred with the pipette to a 100mL conical flask, followed by 3-4 drops of Methyl Orange indicator. This was repeated for another 2-4 conical flasks to use in succession when titrating.
These aliquots were then titrated with the HCl solution from the burette, with care to be taken to swirl the flask quickly throughout to ensure that the flask's contents were as homogenous as possible. When the centre of the flask began to appear pink despite the swirling motion of the flask, the rate of HCl volume added was reduced to a drop by drop feed until the entire solution turned "salmon pink". The volume of HCl used was recorded before the burette was refilled and the titration process repeated for each of conical flasks until three readings were obtained all within 0.1mL of each other for concordant titres. Using these titres of HCl, the concentration of the HCl solution was calculated.
Analysis of cloudy ammonia
In preparation for the dilution of the cloudy ammonia solution, a 20mL pipette was rinsed with distilled water followed by another rinsing with the cloudy ammonia solution before use. Similarly, the 250mL volumetric flask was rinsed with distilled water.
To dilute a sample of the cloudy ammonia product, the bottle was shaken to ensure a homogenous mixture of which 20mL was transferred using a pipette to a 250mL volumetric flask and diluted with distilled water up to the mark on the flask. The volumetric flask was then stopped with a lid and inverted a few times to ensure that the diluted cloudy ammonia solution inside was thoroughly mixed.
20mL aliquots of the diluted cloudy ammonia were transferred to each of the 100mL conical flasks, along with 3-4 drops of methyl orange indicator. The burette of HCl was refilled to the zero mark and administered to one of the conical flasks until the solution turned "salmon pink". The burette was then refilled to the zero mark with HCl standard solution and the titration process was repeated for each of the conical flasks until three concordant titres were obtained.
Using the titre volumes of HCl from the titration and the known volume quantity of the diluted cloudy ammonia solution, the concentration of ammonia was then calculated.
A mass of approximately 1.06g was weighed and measured of anhydrous Na₂CO₃ and diluted to a volume of 250mL in a volumetric flask which was then inverted a number of times to mix. The concentration of this standard solution was then calculated to be used in the titration against HCl.
Standardisation of HCl solution
Before titration, the burette was rinsed with distilled water followed by another rinse with the HCl solution to ensure that the burette was clean and free of impurities as much as possible. This was then filled with the HCl solution.
Similarly, the pipette to be used to transfer aliquots of Na₂CO₃ was rinsed once with distilled water from a 50mL beaker and deposited down the sink before rinsing again with a small sample of Na₂CO₃ solution from another 50mL beaker. Once the pipette had been rinsed, a 20.0mL aliquot of Na₂CO₃ was transferred with the pipette to a 100mL conical flask, followed by 3-4 drops of Methyl Orange indicator. This was repeated for another 2-4 conical flasks to use in succession when titrating.
These aliquots were then titrated with the HCl solution from the burette, with care to be taken to swirl the flask quickly throughout to ensure that the flask's contents were as homogenous as possible. When the centre of the flask began to appear pink despite the swirling motion of the flask, the rate of HCl volume added was reduced to a drop by drop feed until the entire solution turned "salmon pink". The volume of HCl used was recorded before the burette was refilled and the titration process repeated for each of conical flasks until three readings were obtained all within 0.1mL of each other for concordant titres. Using these titres of HCl, the concentration of the HCl solution was calculated.
Analysis of cloudy ammonia
In preparation for the dilution of the cloudy ammonia solution, a 20mL pipette was rinsed with distilled water followed by another rinsing with the cloudy ammonia solution before use. Similarly, the 250mL volumetric flask was rinsed with distilled water.
To dilute a sample of the cloudy ammonia product, the bottle was shaken to ensure a homogenous mixture of which 20mL was transferred using a pipette to a 250mL volumetric flask and diluted with distilled water up to the mark on the flask. The volumetric flask was then stopped with a lid and inverted a few times to ensure that the diluted cloudy ammonia solution inside was thoroughly mixed.
20mL aliquots of the diluted cloudy ammonia were transferred to each of the 100mL conical flasks, along with 3-4 drops of methyl orange indicator. The burette of HCl was refilled to the zero mark and administered to one of the conical flasks until the solution turned "salmon pink". The burette was then refilled to the zero mark with HCl standard solution and the titration process was repeated for each of the conical flasks until three concordant titres were obtained.
Using the titre volumes of HCl from the titration and the known volume quantity of the diluted cloudy ammonia solution, the concentration of ammonia was then calculated.
RESULTS
|
Table 1: Na₂CO₃ standard solution
Created with the HTML Table Generator |
Table 2: Titration of Na₂CO₃ vs HCl solution
Created with the HTML Table Generator |
Table 3: Titration of HCl vs NH₃ solution
Created with the HTML Table Generator |
Analysis
Sodium Carbonate Standard
V (Na₂CO₃) = 250mL
m (Na₂CO₃) = 1.06g
To calculate the concentration of the standard solution, the moles of sodium carbonate will need to be determined to use the formula c = n/V
M (Na₂CO₃) = 106g/mol
n (Na₂CO₃) = m/M
= 1.06g / 106g/mol
= 0.0100 mol
c (Na₂CO₃) = n / V
= 0.0100 mol / 0.250L
= 0.0400M
Concentration of HCl
c (Na₂CO₃) = 0.0400M
V (Na₂CO₃) = 20mL
= 0.020L
∴n (Na₂CO₃ in aliquot) = cV
= 0.0400M x 0.020L
= 8.00x10⁻⁴ mol
V (HCl average titre) = (15.2mL + 15.5mL + 15.5mL) ÷ 3
= 15.4mL
= 0.0154L
c (HCl) = n / V
To find the concentration of HCl, the number of moles of HCl are required which can be determined through the use of the mole ratio from the balanced chemical equation:
(1)Na₂CO₃ (aq) + 2HCl → 2NaCl (aq) + CO₂ (g) + H₂O (l)
Therefore for every unit of Na₂CO₃, there are twice as many molecules of HCl:
n (Na₂CO₃) / 1 = n (HCl) / 2
∴ n (HCl) = 2n (Na₂CO₃)
= 2 x 8.00 x 10⁻⁴ mol
= 1.600 x 10⁻³ mol
∴ c (HCl) = n/V
= 1.600 x 10⁻³ mol / (0.0154L
= 0.104M
Concentration of NH₃
c (HCl) = 0.104M
V(HCl average titre) = (18.8mL + 18.6mL + 18.7mL) / 3
= 18.7mL
= 0.0187L
∴ n (HCl) = cV
= 0.104M x 0.0187L
= 1.94 x 10⁻³ mol
V (NH₃ aliquot) = 20mL
= 0.020L
c (NH₃) = n / V
To determine the concentration of the ammonia, the number of moles of NH₃ are required which can be determined through the use of the mole ratio of the balanced equation:
NH₃ (aq) + HCl (aq) → NH₄⁺ (aq) + Cl⁻ (aq)
Therefore the ratio is 1:1; for every molecule of NH₃ there is one molecule of HCl.
n (NH₃) = n (HCl)
= 1.94 x 10⁻³ mol
∴ c (NH₃) = n / V
= 1.94 x 10⁻³ mol / 0.020L
= 0.097M
To account for the dilution of 20mL sample cloudy ammonia up to 250mL, the original concentration is determined via:
c₁V₁ = c₂V₂ where 1 = initial, and 2 = final
∴c₁ = c₂V₂ / V₁
c₁ (NH₃) = 0.097M x 0.250L / 0.020L
= 1.21M
To compare with the 20g/L claim, the molarity is converted to grams per litre:
n (NH₃) = cV
= 1.21M x 1 L
= 1.21 mol
M (NH₃) = 17g/mol
∴ m (NH₃) = nM
= 1.21 mol x 17g/mol
= 20.6g
= 20.6g
Percent Difference
% Difference = [experimental - theoretical] / [theoretical] x 100%
^^ note absolute difference brackets above
% Difference = (20.6g/L - 20g/L) / (20g/L) x 100%
= 3%
Since experimental result > theoretical result, the experiment was 3% greater than expected.
V (Na₂CO₃) = 250mL
m (Na₂CO₃) = 1.06g
To calculate the concentration of the standard solution, the moles of sodium carbonate will need to be determined to use the formula c = n/V
M (Na₂CO₃) = 106g/mol
n (Na₂CO₃) = m/M
= 1.06g / 106g/mol
= 0.0100 mol
c (Na₂CO₃) = n / V
= 0.0100 mol / 0.250L
= 0.0400M
Concentration of HCl
c (Na₂CO₃) = 0.0400M
V (Na₂CO₃) = 20mL
= 0.020L
∴n (Na₂CO₃ in aliquot) = cV
= 0.0400M x 0.020L
= 8.00x10⁻⁴ mol
V (HCl average titre) = (15.2mL + 15.5mL + 15.5mL) ÷ 3
= 15.4mL
= 0.0154L
c (HCl) = n / V
To find the concentration of HCl, the number of moles of HCl are required which can be determined through the use of the mole ratio from the balanced chemical equation:
(1)Na₂CO₃ (aq) + 2HCl → 2NaCl (aq) + CO₂ (g) + H₂O (l)
Therefore for every unit of Na₂CO₃, there are twice as many molecules of HCl:
n (Na₂CO₃) / 1 = n (HCl) / 2
∴ n (HCl) = 2n (Na₂CO₃)
= 2 x 8.00 x 10⁻⁴ mol
= 1.600 x 10⁻³ mol
∴ c (HCl) = n/V
= 1.600 x 10⁻³ mol / (0.0154L
= 0.104M
Concentration of NH₃
c (HCl) = 0.104M
V(HCl average titre) = (18.8mL + 18.6mL + 18.7mL) / 3
= 18.7mL
= 0.0187L
∴ n (HCl) = cV
= 0.104M x 0.0187L
= 1.94 x 10⁻³ mol
V (NH₃ aliquot) = 20mL
= 0.020L
c (NH₃) = n / V
To determine the concentration of the ammonia, the number of moles of NH₃ are required which can be determined through the use of the mole ratio of the balanced equation:
NH₃ (aq) + HCl (aq) → NH₄⁺ (aq) + Cl⁻ (aq)
Therefore the ratio is 1:1; for every molecule of NH₃ there is one molecule of HCl.
n (NH₃) = n (HCl)
= 1.94 x 10⁻³ mol
∴ c (NH₃) = n / V
= 1.94 x 10⁻³ mol / 0.020L
= 0.097M
To account for the dilution of 20mL sample cloudy ammonia up to 250mL, the original concentration is determined via:
c₁V₁ = c₂V₂ where 1 = initial, and 2 = final
∴c₁ = c₂V₂ / V₁
c₁ (NH₃) = 0.097M x 0.250L / 0.020L
= 1.21M
To compare with the 20g/L claim, the molarity is converted to grams per litre:
n (NH₃) = cV
= 1.21M x 1 L
= 1.21 mol
M (NH₃) = 17g/mol
∴ m (NH₃) = nM
= 1.21 mol x 17g/mol
= 20.6g
= 20.6g
Percent Difference
% Difference = [experimental - theoretical] / [theoretical] x 100%
^^ note absolute difference brackets above
% Difference = (20.6g/L - 20g/L) / (20g/L) x 100%
= 3%
Since experimental result > theoretical result, the experiment was 3% greater than expected.
Table 4: Comparison of ammonia content
| Claimed NH₃ content (g/L) | Experimental NH₃ content (g/L) | Difference (%) |
|---|---|---|
| 20 | 20.6 ±0.536 | 3 ± 2.604 |
DISCUSSION
The ammonia content results from table 4 indicate that the experimental content was slightly greater than the claim on the label; 20.6g/L compared to 20g/L. The significance of these differences was evaluated to be within 3% of the advertised content. This is an acceptable deviation from the advertised content for the products use as a cleaning agent that may not require as stringent tolerances as medicinal or nutritional products.
The uncertainty of the experimental result of ±2.604% (note to students - these calculations were removed from this report for simplicity, but read on how often it is your technique or process that contributes the most to your errors rather than the precision of the equipment you have used) was affected most by the range of titres in the Standardisation of HCl solution procedure where time constraints limited the number of trials possible to obtain concordant titres; that is 3 trials whose volume measurements fall within a 0.1mL range. These titres accounted for 0.97% of the uncertainty, whereas the titres of the Analysis of Cloudy Ammonia procedure were more precise with titres within a ±0.1mL range resulting in an uncertainty of 0.5%. Lastly, the weight measurement of the anhydrous sodium carbonate made the third most significant contribution to the experimental uncertainty of 0.5%. This could be reduced in the future with the use of a three-decimal place weighing balance, using a larger mass and volume to create the Na₂CO₃ standard solution, or using an alternative alkaline standard solution with a higher molecular weight than Na₂CO₃, as this will reduce the relative uncertainties of these measurements.
The ±2.604% uncertainty was not largely due to the measurement equipment utilised in this experiment. The 20mL pipette had a relative uncertainty of 0.15%, and the 250mL volumetric flask had a relative uncertainty of 0.097% provided very precise measurements of volume, however as discussed above, the majority of the uncertainty arose from the large range of titres and the small amounts of Na₂CO₃ measured relative to the number of decimal places that the weighing balance provided.
Anomalies in the titre volumes that did not fall within the concordant values range were discarded, as they were likely to have been affected by the increased rate at which the burette tap was flowing into the conical flask. First trials are often conducted quickly to obtain a ballpark titre volume which can then be anticipated in subsequent trials with greater care once the burette has reached near this ballpark titre volume range and so the first couple of trials are discarded as anomalies.
Future suggestions of improvements to the procedure of this experiment, besides improvements of execution involving greater number of trials, would be to use tris-(hydroxymethyl)aminomethane as the primary standard in the standard solution procedure as it has a greater molecular weight than sodium carbonate; 121.14g/mol vs 105.99g/mol respectively. The process of standardising the HCl solution with Na₂CO₃ was effective in determining the concentration with high precision before using the HCl solution as a standard solution against the cloudy ammonia. The dilution of the original cloudy ammonia solution enabled the titre volumes to fall within an acceptable range to minimise relative errors, and hence increase the precision of the data recorded. For example, if the concentrated form of cloudy ammonia had been used in the aliquot, then the titre volumes of HCl solution would have been approximately 233mL (see working below), which would require the burette to be refilled 5 times to reach the end point of one trial at the same time as accumulating uncertainties in the number of measurements taken per recorded volume.
c (NH₃) = 1.21M
V (NH₃) = 0.020L
n = CV = 1.21M * 0.020L
= 0.0242 mol
n (NH₃) = n (HCl) by mole ratio
∴ n (HCl) = 0.0242 mol
c (HCl) = 0.104M
V = n/c
= 0.0242 mol /0.104M
= 0.233L
= 233mL
*Normally this sort of side calculation would be put in the appendix of the practical report.
The uncertainty of the experimental result of ±2.604% (note to students - these calculations were removed from this report for simplicity, but read on how often it is your technique or process that contributes the most to your errors rather than the precision of the equipment you have used) was affected most by the range of titres in the Standardisation of HCl solution procedure where time constraints limited the number of trials possible to obtain concordant titres; that is 3 trials whose volume measurements fall within a 0.1mL range. These titres accounted for 0.97% of the uncertainty, whereas the titres of the Analysis of Cloudy Ammonia procedure were more precise with titres within a ±0.1mL range resulting in an uncertainty of 0.5%. Lastly, the weight measurement of the anhydrous sodium carbonate made the third most significant contribution to the experimental uncertainty of 0.5%. This could be reduced in the future with the use of a three-decimal place weighing balance, using a larger mass and volume to create the Na₂CO₃ standard solution, or using an alternative alkaline standard solution with a higher molecular weight than Na₂CO₃, as this will reduce the relative uncertainties of these measurements.
The ±2.604% uncertainty was not largely due to the measurement equipment utilised in this experiment. The 20mL pipette had a relative uncertainty of 0.15%, and the 250mL volumetric flask had a relative uncertainty of 0.097% provided very precise measurements of volume, however as discussed above, the majority of the uncertainty arose from the large range of titres and the small amounts of Na₂CO₃ measured relative to the number of decimal places that the weighing balance provided.
Anomalies in the titre volumes that did not fall within the concordant values range were discarded, as they were likely to have been affected by the increased rate at which the burette tap was flowing into the conical flask. First trials are often conducted quickly to obtain a ballpark titre volume which can then be anticipated in subsequent trials with greater care once the burette has reached near this ballpark titre volume range and so the first couple of trials are discarded as anomalies.
Future suggestions of improvements to the procedure of this experiment, besides improvements of execution involving greater number of trials, would be to use tris-(hydroxymethyl)aminomethane as the primary standard in the standard solution procedure as it has a greater molecular weight than sodium carbonate; 121.14g/mol vs 105.99g/mol respectively. The process of standardising the HCl solution with Na₂CO₃ was effective in determining the concentration with high precision before using the HCl solution as a standard solution against the cloudy ammonia. The dilution of the original cloudy ammonia solution enabled the titre volumes to fall within an acceptable range to minimise relative errors, and hence increase the precision of the data recorded. For example, if the concentrated form of cloudy ammonia had been used in the aliquot, then the titre volumes of HCl solution would have been approximately 233mL (see working below), which would require the burette to be refilled 5 times to reach the end point of one trial at the same time as accumulating uncertainties in the number of measurements taken per recorded volume.
c (NH₃) = 1.21M
V (NH₃) = 0.020L
n = CV = 1.21M * 0.020L
= 0.0242 mol
n (NH₃) = n (HCl) by mole ratio
∴ n (HCl) = 0.0242 mol
c (HCl) = 0.104M
V = n/c
= 0.0242 mol /0.104M
= 0.233L
= 233mL
*Normally this sort of side calculation would be put in the appendix of the practical report.
Conclusion
The aim of the investigation was to verify the 20g/L of ammonia claim on the label of 'Cloudy Ammonia'. The experimental results determined a 20.6g/L content with an uncertainty of 2.604%, which differed by a 3% margin from the claim. This minor difference in ammonia content provides evidence of the authenticity of the claim as well as some indication of the tolerances of the product's manufacturing.
References
Clark, J. 2013. Acid-Base Indicators, Chemguide, accessed 03 Sept 2013 from <http://www.chemguide.co.uk/physical/acidbaseeqia/indicators.html#top>.
Clark, J 2013. pH Curves (Titration Curves), Chemguide, accessed 03 Sept 2013 from: <http://www.chemguide.co.uk/physical/acidbaseeqia/phcurves.html>.
Clark, J 2013. pH Curves (Titration Curves), Chemguide, accessed 03 Sept 2013 from: <http://www.chemguide.co.uk/physical/acidbaseeqia/phcurves.html>.